Our MedTech services combine advisory and deep engineering expertise to create clarity and confidence at every stage of product development.


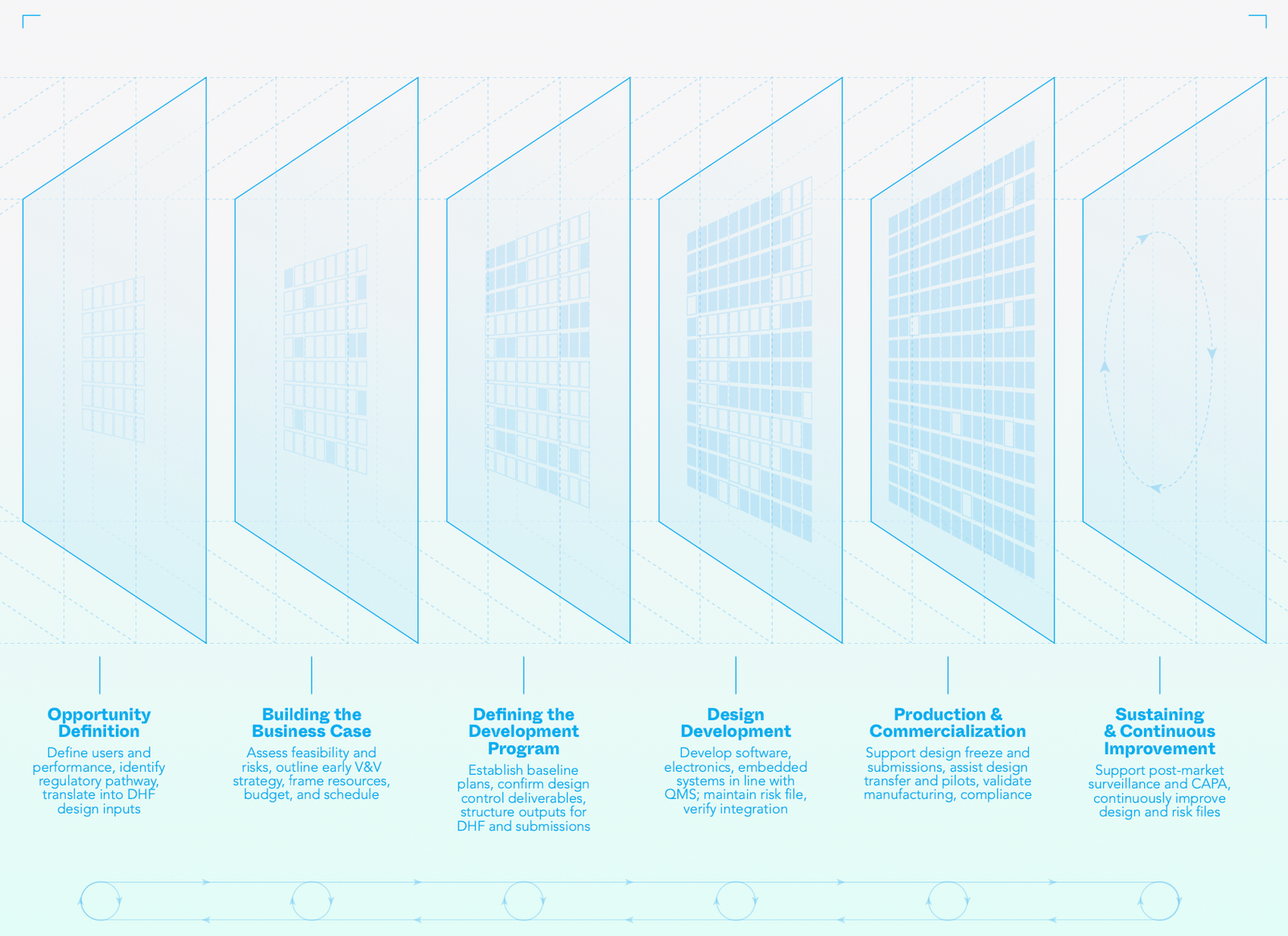
Medical Device Development Process
Medical device commercialisation is complex, but it doesn’t need to be overwhelming. Exa’s development process manages risk from the outset, embeds compliance, and guides you step-by-step to deliver safe, effective, healthcare products.
At every stage of development, our work is viewed through four lenses:
Commercial
Technical
Sustainability
Culture & Alignment
Advisory at the heart of every project
From the first conversation, we help you frame the right problems, test assumptions, and reduce risk before major investment.
Our team provides:
- Program strategy and planning guidance
- Risk identification and mitigation support aligned with ISO 14971
- Feasibility assessments and regulatory insights across global markets
Our approach enables our MedTech partners to progress faster, reduce costs, and increase commercial success.

A fresh approach to product development
As a Certified B Corporation, Exa blends rigorous engineering with empathy, curiosity, and collaboration. We guide every project through four critical lenses: commercial, technical, cultural, and sustainability, to create projects that not only work, but improve patient outcomes and strengthen healthcare systems.
Our Engineers bring proven experience in:
- Electronics, mechanical, firmware, and software development
- Embedded and cloud-connected systems
- Design for Manufacture (DFM) and Design for Verification (DFV)
- Compliance with ISO 13485, IEC 62304, IEC 60601-1 and related standards
The result: medical devices and digital health solutions that are innovative, manufacturable, and regulatory-ready.















